Decorated nanoparticles prevent allergic reactions
New approach prevented anaphylaxis without causing side effects in mouse study
- Link to: Northwestern Now Story
- Besides avoiding allergens, no methods exist to prevent allergic reactions
- Nanotherapy selectively targets immune cells responsible for allergic responses
- Nanoparticle is coated with antibodies that engage with and then deactivate specific cells responsible for responding to a specific allergen
- In a pilot study, therapy prevented anaphylaxis in all mice exposed to an allergen
EVANSTON, Ill. — Northwestern University researchers have developed the first selective therapy to prevent allergic reactions, which can range in severity from itchy hives and watery eyes to trouble breathing and even death.
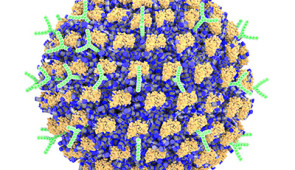
To develop the new therapy, researchers decorated nanoparticles with antibodies capable of shutting down specific immune cells (called mast cells) responsible for allergic responses. The nanoparticle also carries an allergen that corresponds to the patient’s specific allergy. If a person is allergic to peanuts, for example, then the nanoparticle carries a peanut protein.
In this two-step approach, the allergen engages the precise mast cells responsible for the specific allergy, and then the antibodies shut down only those cells. This highly targeted approach enables the therapy to selectively prevent specific allergies without suppressing the entire immune system.
In a mouse study, the therapy demonstrated 100% success in preventing allergic responses without causing noticeable side effects.
The research was published today (Jan. 16) in the journal Nature Nanotechnology. It marks the first nanotherapy for inhibiting mast cells, thus preventing an allergic response to a specific allergen.
“Currently, there are no methods available to specifically target mast cells,” said Northwestern’s Evan A. Scott, who led the study. “All we have are medications like antihistamines to treat symptoms, and those don’t prevent allergies. They counteract effects of histamines after the mast cells already have been activated. If we had a way to inactivate the mast cells that respond to specific allergens, then we could stop dangerous immune responses in severe situations like anaphylaxis as well as less serious responses like seasonal allergies.”
“The biggest unmet need is in anaphylaxis, which can be life-threatening,” said Northwestern’s Dr. Bruce Bochner, an allergy expert and study co-author. “Certain forms of oral immunotherapy might be helpful in some cases, but we currently don’t have any FDA-approved treatment options that consistently prevent such reactions other than avoiding the offending food or agent. Otherwise, treatments like epinephrine are given to treat severe reactions — not prevent them. Wouldn’t it be great if there was a safe and effective treatment for food allergy that consistently made it possible to reintroduce a food into the diet that you used to have to strictly avoid?”
Scott is the Kay Davis Professor of Biomedical Engineering at Northwestern’s McCormick School of Engineering and a member of the Simpson Querrey Institute for BioNanotechnology, of the International Institute for Nanotechnology, of the Chemistry of Life Processes Institute and of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University. Bochner is the Samuel M. Feinberg Emeritus Professor of Medicine (allergy and immunology) at Northwestern University Feinberg School of Medicine. The paper’s first author is Fanfan Du, a postdoctoral fellow in Scott’s laboratory, who worked closely with co-first authors Clayton Rische, a Ph.D. candidate co-mentored by both Bochner and Scott, and Yang Li, a Ph.D. candidate in the Scott lab.
Tricky target
Located in nearly all tissues throughout the human body, mast cells are best known for being primarily responsible for allergic responses. But they also play several other important roles, including regulation of blood flow and fighting parasites. Therefore, fully eliminating mast cells to prevent allergic reactions could be damaging to other useful, healthy responses.
“Although some drugs are under development, there are currently no FDA-approved drugs that inhibit, or eliminate, mast cells,” Bochner said. “This has been difficult mainly because drugs that can affect mast cell activation or survival also target cells other than mast cells, and thus tend to have unwanted side effects due to influences on other cells.”
In previous work, Bochner identified Siglec-6, a unique inhibitory receptor that is highly and selectively found on mast cells. If researchers could target that receptor with an antibody, then they could selectively inhibit mast cells to prevent allergy. But introducing this antibody by itself fell short.
“It was difficult to get a high-enough concentration of the antibody to have an effect,” Scott said. “We wondered if we could enhance this concentration using a nanoparticle. If we could pack a high density of antibodies onto a nanoparticle, then we could make it practical for use.”
Sticking antibodies onto a particle
To pack the antibodies onto a nanoparticle, Scott and his team had to overcome another challenge. For proteins (like antibodies) to stick to a nanoparticle, they typically must form a chemical bond that unfolds (or denatures) the protein, affecting its biological activity. To bypass this challenge, Scott turned to a nanoparticle previously developed in his laboratory.
Unlike more standard nanoparticles that have stable surfaces, Scott’s newly developed nanoparticle comprises dynamic polymer chains, which can independently flip their orientation upon exposure to different solvents and proteins. When put into liquid solutions, the chains orient themselves to achieve favorable electrostatic interactions with water molecules. But when a protein touches the nanoparticle surface, the specific tiny polymer chains at the interface flip their orientations to stably hold onto the protein without covalently bonding to it. Scott’s team also found that water-repelling pockets on protein surfaces were key to the stable interaction.
When binding to surfaces, proteins typically denature, losing their bioactivity. A unique aspect of Scott’s nanoparticles is that they can stably bind enzymes and antibodies while maintaining their 3D structure and biological functions. This means the anti-Siglec-6 antibodies maintained their strong affinity for the mast cell receptors — even when attached to the nanoparticle surfaces.
“This is a uniquely dynamic surface,” Scott said. “Instead of a standard stable surface, it can switch its surface chemistry. It’s made of tiny polymer chains of compounds, which can flip their orientation to maximize favorable interactions with both water and proteins as necessary.”
When Scott’s team mixed the nanoparticles with antibodies, close to 100% of the antibodies successfully attached to the nanoparticles without losing their ability to bind to their specific targets. This resulted in a nanoparticle-based therapy employing surfaces with densely packed and highly controllable amounts of multiple distinct antibodies to target mast cells.
Selective shut down
In order for someone to become allergic, their mast cells capture and display antibodies, specifically immunoglobulin E (IgE) antibodies, for that specific allergen. This enables the mast cells to recognize — and react to — the same allergen upon re-exposure.
“If you have a peanut allergy and have had a response to peanuts in the past, then your immune cells made IgE antibodies against peanut proteins, and the mast cells collected them,” Scott said. “Now, they are waiting for you to eat another peanut. When you do, they can respond within minutes, and if the response is strong enough, it can result in anaphylaxis.”
To selectively target mast cells to respond to a particular allergen, the researchers designed their therapy to engage only mast cells carrying IgE antibodies for that allergen. The nanoparticle uses a protein allergen to engage with IgE antibodies on the mast cells and then uses an antibody to engage the Siglec-6 receptor to shut down the mast cell’s ability to react. And because only mast cells display Siglec-6 receptors, the nanoparticle cannot bind to other cell types — a strategy that effectively limits side effects.
“You can use any allergen that you want, and you will selectively shut down the response to that allergen,” Scott said. “The allergen would normally activate the mast cell. But at the same time the allergen binds, the antibody on the nanoparticle also engages the inhibitory Siglec-6 receptor. Given these two contradictory signals, the mast cell decides that it shouldn’t activate and should leave that allergen alone. It selectively stops a response to a specific allergen. The beauty of this approach is that it does not require killing or eliminating all the mast cells. And, from a safety standpoint, if the nanoparticle accidentally attaches to the wrong cell type, that cell just won’t respond.”
Preventing anaphylaxis in mice
After demonstrating success in cellular cultures using human tissue-derived mast cells, the researchers moved their therapy into a humanized mouse model. Because mast cells in mice do not have the Siglec-6 receptor, Bochner’s team developed a mouse model with human mast cells in their tissues. The researchers exposed the mice to an allergen and delivered the nanotherapy at the same time.
No mice experienced anaphylactic shock and all survived.
“The simplest way to monitor an allergic response is to track changes in body temperature,” Scott said. “We saw no changes in temperature. There was no response. Also, the mice remained healthy and did not display any outward signs of an allergic reaction.”
“Mouse mast cells do not have Siglec-6 on their surface like in humans, but we got as close as we could for now to actual human studies by testing these nanoparticles in special mice that had human mast cells in their tissues,” Bochner said. “We were able to show that these humanized mice were protected from anaphylaxis.”
Next, the researchers plan to explore their nanotherapy for treating other mast cell-related diseases, including mastocytosis, a rare form of mast cell cancer. They also are investigating approaches to loading drugs inside the nanoparticles to selectively kill mast cells in mastocytosis without injuring other cell types.
The study, “Controlled adsorption of multiple bioactive proteins enables targeted mast cell nanotherapy,” was supported by the National Institute of Biomedical Imaging and Bioengineering (grant number 1R01EB030629-01A1) and the National Institute of Allergy and Infectious Disease (grant number R21AI159586).
Multimedia Downloads
Nanoparticle images
Please credit all images to Evan A. Scott/Northwestern University
Interview the Experts