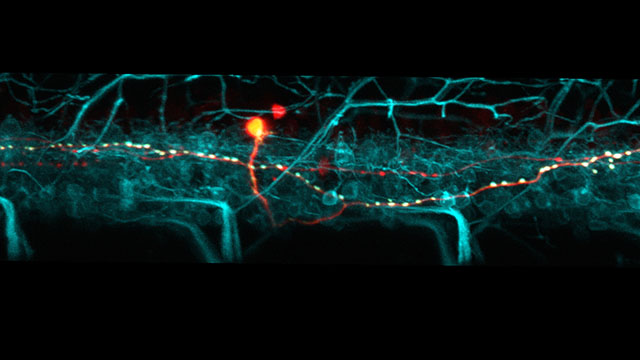
Movement in animals is complex, especially in human beings. A variety of neurons coordinate the activity and inactivity of our muscles, and without that coordination we’d fall flat on our face when we went for a walk.
Scientists have known for quite some time how motor neurons in the spinal cord that activate muscles are organized to generate more forceful output, to transition from walking to jogging to running, for example. Little is known, however, about how spinal inhibitory interneurons work to inhibit, or silence, other neurons and related muscle groups in coordination with the active muscle groups across changing speeds.
Now a Northwestern University research team has discovered in a study of zebrafish that there is a very orderly relationship between when these critical inhibitory neurons are born, their participation in different speeds of movement and what part of a motor neuron they innervate. As a result of this compartmental setup, the amounts of inhibition received by the motor neurons are tuned to different speeds of movement.
Zebrafish is a model organism whose spinal cord works in a fashion similar to our own. Leaning more about the undulating movement of the swimming fish will allow scientists to better understand how humans walk.
The study will be published Oct. 23 by the journal Science.
“A better understanding of how circuits in the spinal cord are organized to coordinate movements puts us in a better position to repair things when damaged or diseased,” said David McLean, corresponding author of the study.
“The fact that we see this pattern in the spinal cord, a relatively primitive part of the nervous system, and in fish, a relatively primitive vertebrate, means that nervous systems made use of this compartmental scheme to regulate activity much earlier than we would have expected,” he said. “It’s a bit like discovering parts for a combustion engine during an archaeological dig.”
It’s a bit like discovering parts for a combustion engine during an archaeological dig.”
McLean is an associate professor of neurobiology in the Weinberg College of Arts and Sciences. He studies the development and functional organization of motor networks. With his research into how these networks control the direction and speed of movements, McLean hopes to reveal broad principles that help explain disorders impacting our capacity to move, such as Parkinson’s disease, epilepsy and spinal injury.
“As neuroscientists, our job is to make sense of the enormous complexity we find in the nervous system that generates behavior,” McLean said. “One way we do that is to look for patterns that give us a sense that there is an underlying logic -- an algorithm that bridges species and is useful in other parts of the brain. This, I think, is one such pattern.”
In their study, McLean and his team used a number of cutting-edge approaches, including using lasers and fluorescent proteins to light up individual neurons and their connections in the transparent fish. They also used electrophysiology in genetically modified fish to characterize function in intact, behaving animals.
The researchers found that inhibitory neurons born first are active during the fastest movements and are most effective at silencing motor output by innervating motor neuron axons, closest to where activity is generated. Next to develop are interneurons active at intermediate speeds that strongly inhibit motor neuron cell bodies, followed by those active at slow speeds that weakly inhibit dendrites.
Collectively, the arrangement of inhibitory inputs in different structural compartments simplifies the process of ensuring appropriate patterns of motor output at different speeds, McLean said, since different cell types can provide a stronger or weaker ‘veto’ over motor output depending on when they are active.
“This compartmental pattern exists in other animals and brain regions, but using zebrafish we discovered clear functional consequences by watching the circuits develop, similar to understanding how a car works by following the assembly line,” said Sandeep Kishore, first author of the study and a research associate in McLean’s lab.
During early development of the zebrafish, inhibitory networks controlling the most forceful movements are the first to become functional, followed by those controlling more subtle, controlled movements.
“This makes some sense when you consider human development, where our first actions are kicking movements in the womb,” McLean said. “Only later do we require and acquire finer motor skills.”
The research was supported by the National Institutes of Health (grants R01 NS067299 and U19 NS104653).
The title of the paper is “Orderly compartmental mapping of premotor inhibition in the developing zebrafish spinal cord.” In addition to McLean and Kishore, other authors are Eli B. Cadoff and Moneeza A. Agha, both of Northwestern.