Implantable ‘living pharmacy’ could control body’s sleep/wake cycles
Project receives DARPA contract worth up to $33 million over four-and-a-half years
- Link to: Northwestern Now Story
- Device will harness the same peptides the the body makes to regulate sleep cycles
- Researchers aim to halve the time it takes to recover from disrupted sleep/wake cycles
- Device could be a powerful tool for military personnel and shift workers, including first responders
EVANSTON, Ill. — A Northwestern University-led team of researchers has signed a cooperative agreement with the Defense Advanced Research Projects Agency (DARPA) to develop a wireless, fully implantable device that will control the body’s circadian clock, halving the time it takes to recover from disrupted sleep/wake cycles.
The first phase of the highly interdisciplinary program will focus on developing the implant. The second phase, contingent on the first, will validate the device. If that milestone is met, then researchers will test the device in human trials, as part of the third phase. The full funding corresponds to $33 million over four-and-a-half years.
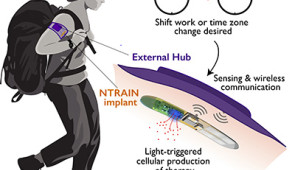
Nicknamed the “living pharmacy,” the device could be a powerful tool for military personnel, who frequently travel across multiple time zones, and shift workers including first responders, who vacillate between overnight and daytime shifts.
Called NTRAIN (Normalizing Timing of Rhythms Across Internal Networks of Circadian Clocks), the project is a part the Advanced Acclimation and Protection Tool for Environmental Readiness (ADAPTER) program, DARPA’s new program to help address the challenges of travel, including jetlag, fatigue and gastrointestinal issues. Circadian clock research will be led by sleep experts at Northwestern’s Center for Sleep and Circadian Biology (CSCB). Synthetic biologists at Rice University will lead cellular engineering efforts. And Northwestern engineers will join researchers from Rice and Carnegie Mellon universities and Blackrock Microsystems to develop bioelectronic components.
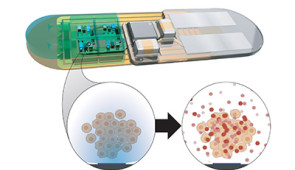
Combining synthetic biology with bioelectronics, the team will engineer cells to produce the same peptides that the body makes to regulate sleep cycles, precisely adjusting timing and dose with bioelectronic controls. When the engineered cells are exposed to light, they will generate precisely dosed peptide therapies.
“This control system allows us to deliver a peptide of interest on demand, directly into the bloodstream,” said Northwestern’s Jonathan Rivnay, principal investigator of the project. “No need to carry drugs, no need to inject therapeutics and — depending on how long we can make the device last — no need to refill the device. It’s like an implantable pharmacy on a chip that never runs out.”
Rivnay is an assistant professor of biomedical engineering in Northwestern’s McCormick School of Engineering. Other Northwestern members of the interdisciplinary team are: professors Fred W. Turek, Martha Hotz Vitaterna, Josiah Hester, Guillermo Ameer, Peng Jiang and Phyllis C. Zee — representing Weinberg College of Arts and Sciences, McCormick and Feinberg School of Medicine.
Beyond controlling circadian rhythms, the researchers believe this technology could be modified to release other types of therapies with precise timing and dosing for potentially treating pain and disease. The DARPA program also will help researchers better understand sleep/wake cycles, in general.
“The experiments carried out in these studies will enable new insights into how internal circadian organization is maintained,” said Turek, who co-leads the sleep team with Vitaterna. “These insights will lead to new therapeutic approaches for sleep disorders as well as many other physiological and mental disorders, including those associated with aging where there is often a spontaneous breakdown in temporal organization.”
Members from other institutions include Doug Weber, Tzahi Cohen-Karni, Darcy Griffin, Carl Olson and Matt Smith at CMU; Karrie Fitzpatrick of the University of Minnesota; Omid Veiseh, Jacob Robinson, Isaac Hilton, Kaiyuan Yang and Caleb Kemere of Rice University; Florian Solzbacher of the University of Utah; and Rob Franklin of Blackrock Microsystems.
Multimedia Downloads
Living pharmacy
Please credit images to Northwestern University
Interview the Experts