For reproduction to be successful, an egg cell must divide perfectly. Egg cell divisions are so error-prone, however, that they are the leading cause of miscarriages and birth defects in humans.
In a new study, Northwestern University researchers discovered a previously unknown mechanism in roundworms that protects their egg cells from division errors. Uncovering and understanding this hidden mechanism could ultimately lead to new strategies for combatting infertility in humans.

“Prior to our work, certain proteins were thought to be essential for cells to divide,” said Northwestern’s Sadie Wignall, who led the study. “However, when we removed these proteins, we were surprised to discover a previously hidden ‘backup’ mechanism that was able to kick in when the main proteins were missing. We uncovered something that other researchers missed because, if the major mechanism is in place, then you wouldn’t know that backup existed.”
The study was published today (March 29) in the journal eLife.
Wignall is an associate professor of molecular biosciences at Northwestern’s Weinberg College of Arts and Sciences. Gabriel Cavin-Meza, a graduate student in Wignall’s laboratory, is the paper’s first author.
When an egg is fertilized with sperm, the resulting embryo begins to rapidly divide, eventually developing into a healthy organism. If either the egg or the sperm have the wrong amount of genetic material, however, then the organism cannot properly develop.
While other cells in the human body divide perfectly more than 99% of the time, egg cells are mysteriously error prone. About 10-25% of the time, egg cells incorrectly divide, resulting in the wrong amount of genetic material ending up in the embryo.
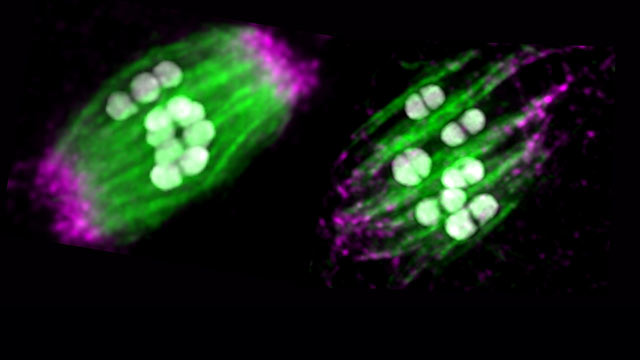
To understand why egg cells are more susceptible to errors, Wignall studies a football-shaped structure, called the spindle, which organizes genetic material before the egg divides.
“The spindle is like a machine,” Wignall said. “It lines up chromosomes and then pulls them apart, ensuring that the right number of chromosomes end up in each half of the cell.”
We uncovered something that other researchers missed because, if the major mechanism is in place, then you wouldn’t know that backup existed.”
molecular bioscientist
The spindle is made up of long ropelike structures called microtubules. The microtubules gather into points at each pole of the spindle, giving it its football shape. Then microtubules attach to the chromosomes and eventually pull them apart.
“Motor proteins bind to the microtubules and take steps to move along them — just like humans walk using their legs,” Wignall said. “When microtubules are first formed, they are a random mess. Then the motors use this walking motion to push the microtubules around to arrange them into the spindle structure.”
Before Wignall’s new study, researchers believed two motor proteins (dynein and kinesin-12) were primarily responsible for this task. But when Cavin-Meza removed both proteins from roundworms’ egg cells, he saw something shocking.
“When we removed these proteins, it made the entire spindle blow apart,” Wignall said. “Then we were surprised to see the spindle reform.”
In the absence of dynein and kinesin-12, another motor protein (called kinesin-5) came out of hiding to perform its backup duty. In the end, this previously unknown mechanism restored the spindle structure, allowing the chromosomes to be pulled apart.
Although this study was completed in C. elegans — a well-studied roundworm that is commonly used as a model for reproduction — Wignall believes similar mechanisms might be present in humans. But because so few human eggs cells are donated to science, researchers study model organisms to probe questions and work out details before examining humans.
“All of the components in our study are also present in human eggs,” Wignall said. “Spindles seem to form the same way in humans and even look exactly the same. It would be really interesting to see if humans also have this backup mechanism.”
The study, “Multiple motors cooperate to establish and maintain acentrosomal spindle bipolarity in C. elegans oocyte meiosis,” was supported by the National Institutes of Health (award numbers R01GM124354 and T32 CA009560).